ELIAS Cancer Immunotherapy (ECI®)
The ELIAS Cancer Immunotherapy (ECI®) combines an autologous vaccine with cell therapy to target a patient’s unique cancer cells. This personalized approach can improve clinical outcomes and provide a better quality of life for canine patients. ECI® is USDA-approved for treatment of canine osteosarcoma and, as an autologous prescription product, can also be used by licensed veterinarians to treat other cancers.
Two-step Treatment. Explained.
ECI® harnesses the molecular machinery within T cells to eliminate cancer that is actively growing. It utilizes a proprietary and patented combination of cancer vaccination pretreatment and activated “killer” T cell immunotherapy.
Because this therapy uses the patient’s unique cancer to produce the vaccine, amputation of the affected limb must not occur prior to beginning treatment.
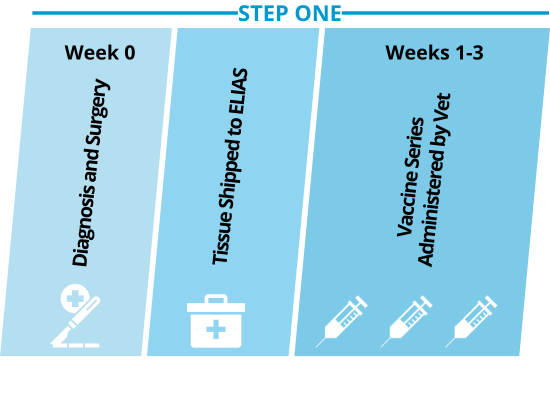
Step One: Personalized vaccines prime the immune system to recognize the dog’s unique cancer.
The primary tumor is surgically removed from the canine patient. This cancer tissue is used to produce a personalized cancer vaccine, which primes the dog’s immune system to recognize the cancer.
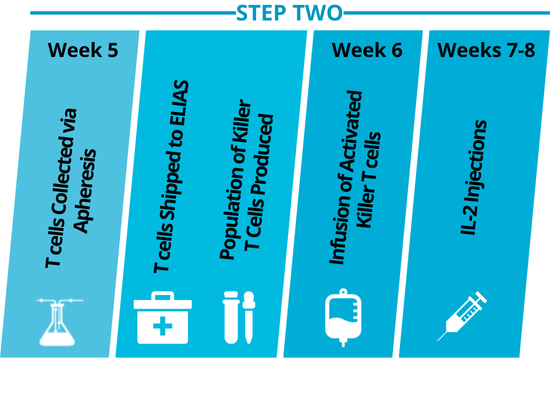
Step Two: T cell activation and reinfusion launches the immune response.
The primed immune cells are collected by apheresis, then activated and expanded into an army of killer T cells. The T cells are reinfused into the dog where they attack cancer cells in the body. Low-dose IL-2 injections further support the immune response.
Immunotherapy. Personalized.
ECI® is unique because of its dual approach to treatment: it brings together the power of a personalized cancer vaccination and activated “killer” T cell therapy to trigger the patient’s own immune system to attack cancer cells.
Watch the video to learn how it works.
Backed by USDA-Reviewed Studies
ECI® is the only USDA-licensed therapy for the treatment of canine osteosarcoma.
ECI® demonstrated a reasonable expectation of efficacy in a multi-site, prospective, randomized, positive-controlled clinical trial. This study also demonstrated that the immunotherapy was well-tolerated, with no reported treatment-related serious adverse reactions.1
SUCCESS STORY
Roscoe: Apparent Disease Regression
Roscoe was diagnosed with osteosarcoma at age 6 in May 2017 and received ECI® treatment following amputation. Several months later, a distant subcutaneous OSA metastasis was discovered, which then resolved without further medical intervention. This suggests that Roscoe’s immune system, boosted by ECI®, remained active against the cancer for a sustained length of time.
“The people at ELIAS Animal Health are my heroes. Roscoe is living a normal life and [didn’t] die of cancer, and for that I will be eternally grateful.” – Roscoe’s Mom
INDICATIONS AND USAGE
ELIAS Cancer Immunotherapy (ECI®) is recommended for treatment of osteosarcoma in dogs. Results may vary between patients.
IMPORTANT SAFETY INFORMATION
Glucocorticoids or any other immunosuppressive drugs are not permitted beginning 14 days prior to surgery until 30 days following end of treatment with ECI®. Immunomodulating drugs, herbal supplements, and other naturopathic therapies are not permitted beginning at surgery until 30 days following T cell infusion. If necessary, dexamethasone is permitted to mitigate serious adverse events following T cell infusion.1
All reported adverse reactions reported in the clinical trial (ECI-OSA-04) were mild to moderate in severity. For additional safety information, please see full prescribing information.1
REFERENCES