ASCENT Study: 18-Month Update
The ASCENT study is evaluating the use of the ELIAS Cancer Immunotherapy (ECI®) in combination with a single dose of chemotherapy to treat bone cancer in dogs. ECI® is an adoptive cell therapy approved by the USDA for treatment of canine osteosarcoma.
The ASCENT study analysis suggests that the cytotoxic effect of chemotherapy may enhance immunological effects of ECI® with significant improvement in survival outcomes.1
1Bryan JN et al. One-dose carboplatin followed by vaccine-enhanced adoptive cell therapy (ECI®) improves outcomes compared to four-dose carboplatin in dogs with osteosarcoma — ASCENT Study 18-month update. Presented at the Veterinary Cancer Society/Veterinary Society of Surgical Oncology Collaborative Conference; March 18–21, 2026; Savannah, GA.
Key Observations
In this analysis1, dogs that received one dose of chemotherapy (carboplatin) followed by ECI® showed longer survival compared to dogs treated with 4-dose carboplatin alone, and the protocol was well-tolerated. These findings support the potential benefit of this combination therapy.


Study Overview
ASCENT is a single-arm, multi-center combination therapy study evaluating outcomes associated with integration of ECI® vaccine-enhanced adoptive cell therapy with chemotherapy following amputation in dogs with appendicular osteosarcoma. Outcomes were compared with matched historical controls treated with four-dose carboplatin.
Combination Protocol Summary
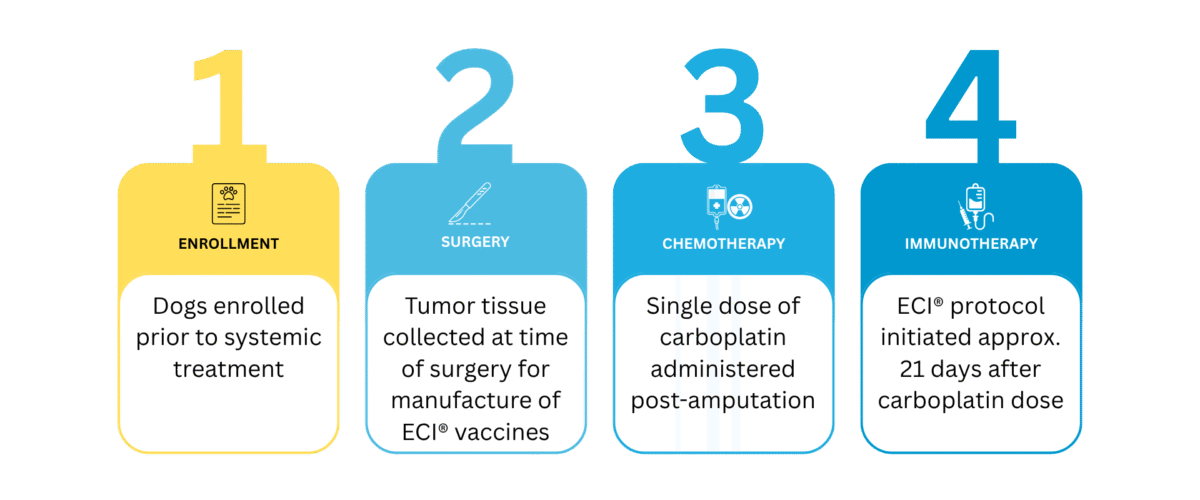
Clinical Context
Canine appendicular osteosarcoma remains associated with a high risk of metastatic progression following local tumor control. Post-amputation chemotherapy protocols are commonly incorporated to address micrometastatic disease risk.
Historically reported median survival times following:
- amputation alone: approximately 2–4 months
- amputation plus four-dose carboplatin: approximately 6–11 months
In human patients, improved outcomes have been reported for those receiving combined therapies, such as chemotherapy, adoptive cell therapy and checkpoint inhibitors.
Relationship to Prior Clinical Evidence
The ASCENT study builds on the results of the BASE study to evaluate the safety and effectiveness of combining ECI® with chemotherapy.
About ECI®
ELIAS Cancer Immunotherapy (ECI®) is an adoptive cell therapy approved by the USDA for treatment of canine osteosarcoma and distributed under an Autologous Prescription Product license.
SCIENTIFIC CITATION
1Bryan JN et al. One-dose carboplatin followed by vaccine-enhanced adoptive cell therapy (ECI®) improves outcomes compared to four-dose carboplatin in dogs with osteosarcoma — ASCENT Study 18-month update. Presented at the Veterinary Cancer Society / Veterinary Society of Surgical Oncology Collaborative Conference; March 18–21, 2026; Savannah, GA.

